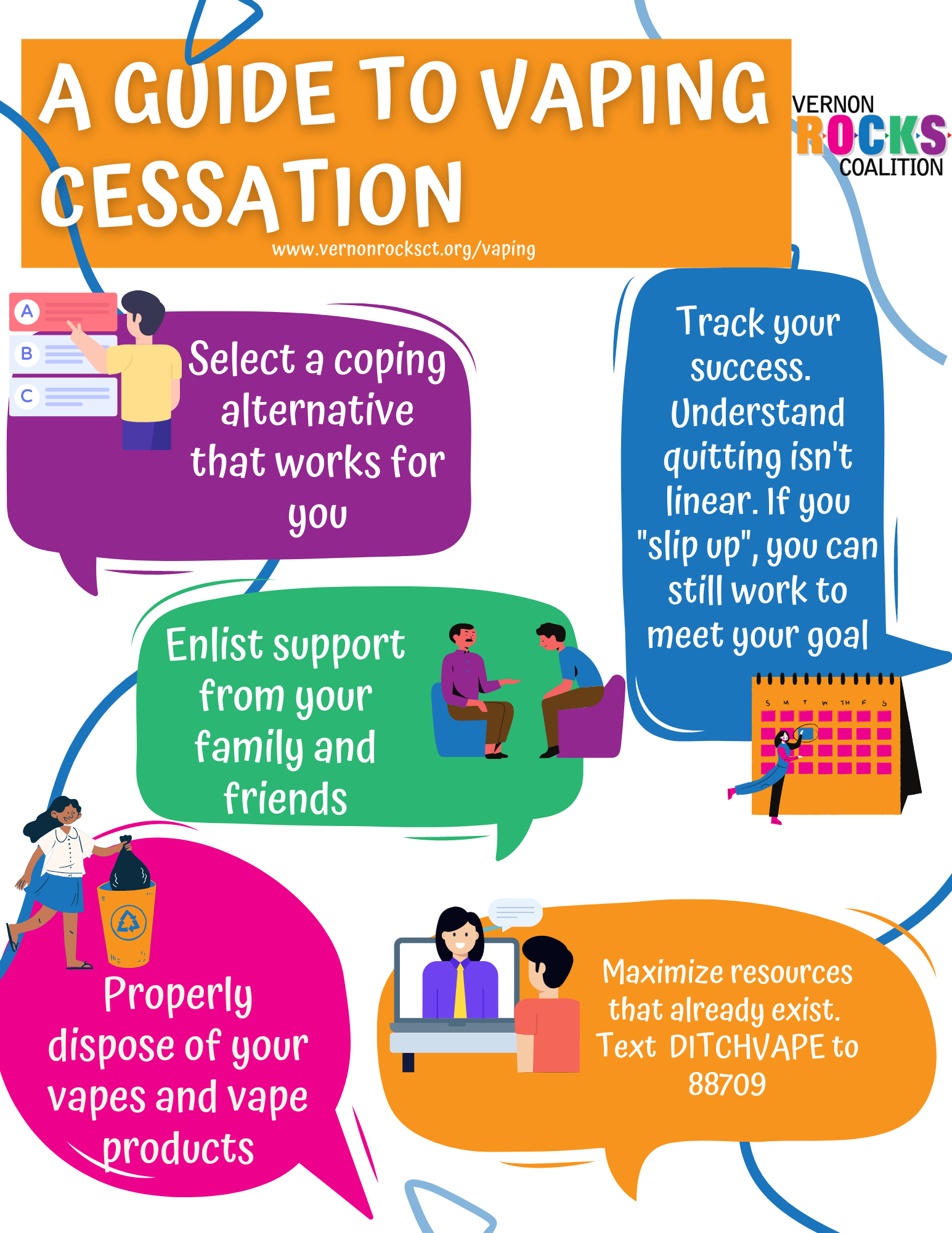
U.S. health innovation is a cornerstone of global biomedical advancements, often hailed as the gold standard in healthcare innovation. This vibrant ecosystem has evolved through significant public-private research partnerships, particularly since World War II when government-supported research laid the groundwork for groundbreaking treatments and technologies. The unique collaboration between academic institutions and federal funding has driven forward-thinking drug development history, yielding life-saving medications and therapeutic breakthroughs. As the demand for efficient healthcare solutions continues to rise, the U.S. remains at the forefront, demonstrating how strategic investment in science can lead to remarkable public health outcomes. The success of U.S. health innovation not only enhances national well-being but also sets a powerful example for countries looking to boost their healthcare systems.
The innovation landscape in U.S. healthcare exemplifies a dynamic interplay of science, technology, and collaboration, reshaping how medical advancements are conceived and delivered. With robust partnerships between government bodies and research institutions, this system thrives on the principles of cooperative exploration and access to crucial federal resources. The priorities of the U.S. health sector reflect an emphasis on addressing urgent health challenges through strategic biomedical R&D initiatives, heralding a new era in therapeutic solutions and drug discovery. This collaborative model fosters not only the emergence of pioneering treatments but also an environment where emerging scientists can develop their talents and contribute to future discoveries. As the world looks toward the U.S. for leadership in healthcare solutions, the importance of nurturing such innovation ecosystems becomes abundantly clear.
The Roots of U.S. Health Innovation
The U.S. health innovation ecosystem has its origins in a time of great need, particularly during World War II. The collaboration between academia and federal agencies initiated a critical response to the challenges posed by the war, yielding significant advancements in medicine and technology. Leaders from universities and research institutions approached President Franklin D. Roosevelt to mobilize scientific talent for military innovations—this marked the inception of a partnership that has continued to evolve since then. By pooling resources and expertise, the government and research institutions catalyzed breakthroughs that would lay the foundation for future healthcare innovations.
This public-private collaboration became essential during the war as the U.S. military faced unprecedented health challenges, where infectious diseases posed a greater threat to soldiers than actual battlefield injuries. The establishment of the Office of Scientific Research and Development (OSRD) exemplified this innovative partnership, facilitating the necessary R&D to develop solutions swiftly. The roots of this initiative underscore the significance of robust cooperation between government and academia in fostering U.S. health innovation, paving the way for modern-day biomedical advancements.
Public-Private Research: A Catalyst for Progress
The interaction between public and private sectors has been crucial for driving change in the U.S. health landscape. The historical context reveals that federal funding has traditionally underpinned significant investments in research and development, particularly in the biomedical sector. This strong partnership has not only propelled drug development history in the U.S. but has also been imitated globally, showcasing its effectiveness. Understanding the dynamic between these sectors helps illuminate how strategies for federal investment can yield real-world health outcomes, benefiting patients and healthcare professionals alike.
However, the balance of this relationship is increasingly becoming a topic of debate. The recent scrutiny over federal funding, particularly regarding proposed caps on reimbursement for indirect costs in biomedical research, raises concerns about the sustainability of public-private partnerships. Maintaining this vital collaboration is essential, as history has shown that productive joint efforts lead to transformative healthcare innovations. As stakeholders navigate these challenges, ensuring continued investment in partnerships will be crucial for the future of U.S. health innovation.
The Role of Federal Funding in Biomedical Advancements
Federal funding has played an instrumental role in shaping the landscape of biomedical advancements in the United States. Historical initiatives, particularly those stemming from World War II, established a framework for sustained investment in health research. Through institutions like the National Institutes of Health (NIH), substantial resources have been allocated to support innovative studies that lead to new therapies and drugs. This funding mechanism not only drives research within academic institutions but also supports corporate endeavors, facilitating a collaborative environment that stimulates biomedical breakthroughs.
In light of contemporary challenges, the discussion surrounding federal funding has become increasingly urgent. Experts argue that cuts to research funding could set back decades of progress made in healthcare innovation. By understanding the implications of these funding dynamics, stakeholders can advocate for policies that ensure the U.S. remains at the forefront of global health advancements. The intricate relationship formed between federal investment and biomedical research underscores the importance of sustained support for future discoveries that will continue to improve patient outcomes.
Innovation Strategies in Drug Development History
The history of drug development in the U.S. is marked by significant innovation strategies that have evolved over the decades. From the early days of trial and error in chemical drug discovery to today’s sophisticated methodologies, the journey has been driven by a need for effective medical solutions. The wartime efforts during World War II exemplified the urgency for rapid advancements, resulting in significant innovations such as the mass production of penicillin. These early efforts laid the groundwork for structured drug development processes and a more scientific approach to therapeutics.
As we moved through the latter half of the 20th century, the pharmaceutical landscape transformed dramatically. Advances in biology and chemistry improved the precision with which new medications could be developed, significantly expanding the potential for treating various diseases. The infrastructure built during the war laid the foundation for the ‘Golden Age’ of drug discovery, where innovative chemistry and biological discoveries merged to produce life-saving medications. Today, this rich history informs current practices and underlines the importance of continued investment in drug development and health innovation.
The Impact of War on Health Innovation
The impact of war on health innovation cannot be overstated; historical data suggests that significant advancements in medical technology and drug development emerged from the urgencies of military conflict. The establishment of the OSRD during World War II streamlined research efforts, provided funding, and coordinated various stakeholders, allowing for rapid innovation. This wartime focus on health-related challenges, including epidemic diseases, catalyzed research that ultimately benefited not just the military but civilian sectors as well.
Moreover, the pressing need for effective medical solutions during wartime laid the groundwork for ongoing partnerships between government, academia, and industry. These alliances have since proved instrumental in maintaining a robust pipeline of health innovations that support both public health and military preparedness. The lessons learned from wartime collaborations continue to shape the U.S. health innovation ecosystem, emphasizing the critical nature of responsive research in times of urgency.
Training the Next Generation of Scientists
Training a new generation of scientists has always been an integral component of the U.S. health innovation landscape. During and after World War II, thousands of young graduates were engaged in research initiatives, receiving hands-on experience that would galvanize their careers. This influx of fresh talent brought innovative ideas and diverse perspectives to the scientific community, helping to drive forward developments in biomedical research and technology. Programs initiated during the wartime efforts ensured that a skilled workforce was prepared to tackle the ongoing and future challenges in health.
Today, the emphasis on scientific training continues to evolve, with a focus on fostering critical thinking and hands-on learning in the fields of biology, technology, and medicine. Universities and research institutions have responded by creating programs that bridge the gap between theoretical knowledge and practical application, ensuring that students graduate ready to contribute meaningfully to the health innovation ecosystem. This infusion of new talent not only supports the research pipeline but also reinforces the commitment to continuous improvement in healthcare outcomes.
Legacy of Public-Private Research Partnerships
The legacy of public-private research partnerships in the U.S. has cultivated a vibrant health innovation ecosystem. These collaborations have not only facilitated groundbreaking discoveries but have also created a robust framework for ongoing research and development. By combining the resources and expertise of both sectors, significant advancements in healthcare have emerged. The success stories from these collaborations illustrate how strategic partnerships can lead to innovative solutions that improve patient care and advance medical technology.
However, the sustainability of these partnerships is facing new challenges in today’s fiscal climate. The debate surrounding federal funding has raised concerns about potentially destabilizing a successful model that has thrived for decades. Advocates for public-private partnerships argue that preserving this collaborative framework is essential for nurturing future healthcare innovations. The legacy established over the years underscores the importance of maintaining these partnerships to ensure continued progress in biomedicine and healthcare on a global scale.
Future Directions of Health Innovation
Looking ahead, the future directions of health innovation hinge on adapting to new challenges and leveraging emerging technologies. As advancements in artificial intelligence, genomics, and data analytics reshape the healthcare landscape, the potential for novel therapeutic solutions increases. The collaborative efforts seen during past crises, such as World War II, remind us of the importance of agile responses to pressing health issues. By reinvigorating public-private partnerships, the U.S. can facilitate the rapid translation of scientific discoveries into real-world health applications.
Ensuring that all stakeholders, including government, industry, and academia, remain engaged in the health innovation ecosystem will be critical for advancing research. The commitment to fostering collaborations that prioritize shared goals will fuel the development of cutting-edge therapies and healthcare solutions. As we face growing public health challenges, maintaining the momentum of innovation will ensure the U.S. continues to lead in the global health innovation space.
Balancing Efficiency and Innovation in the System
As the U.S. health innovation system continues to evolve, the challenge lies in balancing efficiency with the need for continued innovation. Historical trends have shown that baseline funding and strategic investments are essential for ensuring the stability of the research ecosystem. Policymakers must prioritize mechanisms that foster collaboration between public institutions and private companies, thereby enhancing overall efficiency without stifling creativity or progress. As seen in the past, a robust system that encourages exploration and experimentation will pay dividends in terms of breakthroughs in healthcare.
However, this challenge is further complicated by the pressures of budget constraints and changing political landscapes. Ensuring that public funding mechanisms remain responsive to the needs of modern research environments is paramount. By refining policies to support indirect costs and incentivize collaborations, we can help sustain the innovation that has characterized the U.S. health system for over 80 years. Ultimately, preserving the delicate balance between efficiency and innovation holds the key to maintaining excellence in healthcare and biomedical advancements.
Frequently Asked Questions
How has U.S. health innovation evolved since World War II?
U.S. health innovation has significantly evolved since World War II, primarily driven by public-private research partnerships. These collaborations emerged from the urgent need for medical advancements during the war, notably in drug development, which led to critical discoveries like penicillin. Since then, the integration of federal funding has allowed academic research to flourish, resulting in groundbreaking biomedical advancements.
What role does federal funding play in U.S. health innovation?
Federal funding is pivotal in U.S. health innovation as it supports academic research and development in the biomedical field. This funding fosters innovation by enabling researchers to explore new ideas, conduct experiments, and ultimately contribute to drug development history. Without substantial federal financial backing, many transformative health innovations might not have materialized.
What are the primary components of the U.S. healthcare innovation ecosystem?
The U.S. healthcare innovation ecosystem comprises three primary components: academic institutions, federal agencies like the National Institutes of Health (NIH), and the life sciences industry. This triadic relationship fosters an environment ripe for healthcare innovation, with public-private research partnerships being a critical element that has spurred biotechnological advancements.
How did public-private research partnerships enhance U.S. health innovation during wartime?
Public-private research partnerships greatly enhanced U.S. health innovation during wartime by mobilizing civilian scientists to address urgent technological challenges. The establishment of the Office of Scientific Research and Development (OSRD) during World War II exemplified this approach, leading to major advancements in drug development and biomedical research that laid the groundwork for modern innovations.
What impact did World War II have on drug development in the U.S.?
World War II had a transformative impact on drug development in the U.S. by catalyzing innovation and establishing infrastructures for biomedical advancements. The war necessitated rapid medical solutions, prompting federal investment in research that led to the successful mass production of antibiotics like penicillin, which dramatically improved soldier health and established a foundation for future drug discoveries.
How do universities contribute to the U.S. health innovation landscape?
Universities contribute to the U.S. health innovation landscape by serving as research hubs where new biomedical technologies are developed. Through collaboration with federal agencies and private industries, they conduct groundbreaking research supported by federal funding, fostering an environment of continuous healthcare innovation and training the next generation of scientists.
What challenges does U.S. health innovation currently face?
Currently, U.S. health innovation faces challenges related to federal funding cuts and regulatory changes that could threaten the sustainability of public-private research partnerships. Moreover, the pressure to maintain efficient use of resources while fostering innovation remains a critical concern, as any significant disruption could impact the nation’s position as a leader in biomedical advancements.
Why is the partnership between federal agencies and private industries critical for health innovation?
The partnership between federal agencies and private industries is critical for health innovation because it combines public funding with private sector efficiency and expertise. This collaboration has historically led to significant breakthroughs in biomedicine, as both sectors bring their strengths to tackle complex health challenges, drive drug development, and promote advancements in healthcare technology.
What historical milestones define the drug development history in the U.S.?
Historical milestones in U.S. drug development history include the mass production of penicillin during World War II, the establishment of rigorous FDA regulations in the mid-20th century, and the antibiotic revolution spurred by wartime research efforts. These milestones underscore the critical impact of public-private research collaborations and federal funding in shaping the pharmaceutical landscape.
| Key Points | Details |
|---|---|
| Origins of U.S. Health Innovation | Initiated during World War II with government-supported research, leading to the mass production of penicillin. |
| Impact of WWII on Research | The war catalyzed the establishment of the OSRD, promoting innovation in medical and technological fields. |
| Public-Private Partnerships | Partnerships between the federal government, universities, and private sectors emerged and thrived. |
| Government Role | Provided essential funding for biomedical research that fostered new drug discoveries and technologies. |
| Challenges and Criticism | Current scrutiny over federal funding and the potential for cuts under new policies. |
| Lasting Impacts | Foundation for postwar biomedical innovations and development of new therapies. |
| Training Future Scientists | Engaged thousands of graduate students, fostering a new generation of researchers. |
| Sustaining Innovation | The existing partnership model remains vital for continuing U.S. leadership in health innovation. |
Summary
The U.S. health innovation sector stands as a global leader, having evolved through decades of strategic partnerships and significant government support. From its origins in World War II, where the urgent need for medical advancements led to breakthroughs like penicillin, the collaboration between federal agencies, universities, and private industries has propelled the country to the forefront of biomedicine. This partnership has not only been critical for health advancements but has also set a framework that many nations aspire to replicate. Looking ahead, preserving and enhancing this innovative ecosystem will be crucial for meeting future healthcare challenges and maintaining the U.S.’s position as a pioneer in health innovation.